 
The elements with the highest ionization energies are generally those with the most negative electron affinities, which are located toward the upper right corner of the periodic table. If this relative attraction is great enough, then the bond is an ionic bond. A polar covalent bond is one in which one atom has a greater attraction for the electrons than the other atom.A nonpolar covalent bond is one in which the electrons are shared equally between two atoms.As demonstrated below, the bond polarity is a useful concept for describing the sharing of electrons between atoms within a covalent bond: For most covalent substances, their bond character falls between these two extremes. For example, while the bonding electron pair is shared equally in the covalent bond in \(Cl_2\), in \(NaCl\) the 3s electron is stripped from the Na atom and is incorporated into the electronic structure of the Cl atom - and the compound is most accurately described as consisting of individual \(Na^ \) and \(Cl^-\) ions (ionic bonding). 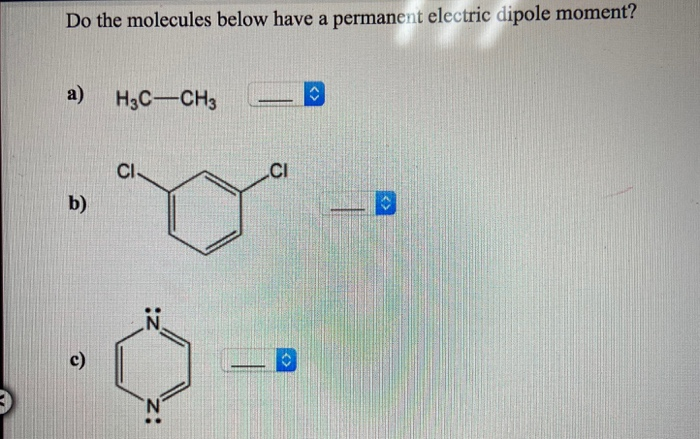
The electron pairs shared between two atoms are not necessarily shared equally. To calculate the percent ionic character of a covalent polar bond.To define electronegativity and bond polarity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
